In results of an ongoing phase III Comirnaty trial with over 2,000 participants aged 12-15 years, vaccine efficacy against symptomatic COVID-19 from 7 days after dose two was 100% (95% CI 78.1-100%) with no cases reported in the vaccine arm. Vaccine efficacy, immunogenicity and effectiveness: There is high level evidence indicating strong immunogenicity and vaccine efficacy against symptomatic COVID-19 in adolescents from clinical trials of Pfizer and Moderna. Evidence of potential acceptance of vaccination in this age group.īenefits of vaccinating adolescents against COVID-19 Direct benefits against COVID-19 in children.Mathematical modelling relating to population level impact of vaccinating this age group.Programmatic implications and the potential delivery strategies to extend vaccination to the young adolescent age group.Safety of COVID-19 vaccines, including risk of myocarditis and pericarditis after receiving mRNA vaccines in adolescents and young adults reported overseas.Epidemiology of COVID-19 in adolescents including disease severity and complications, and their role in transmission in the population.Safety, efficacy and effectiveness of COVID-19 vaccines in adolescents from clinical trials and overseas vaccination programs.The Therapeutic Goods Administration (TGA) provisional registration of Pfizer was extended on 23 July to include all people from 12 years of age and above in a two-dose schedule, and on 4 September Moderna was provisionally registered for use in 12 to 17 year old adolescents.ĪTAGI has developed these current recommendations for all individuals aged 12 years and above by carefully considering the relevant benefits, risks, uncertainties and evidence on the following: 
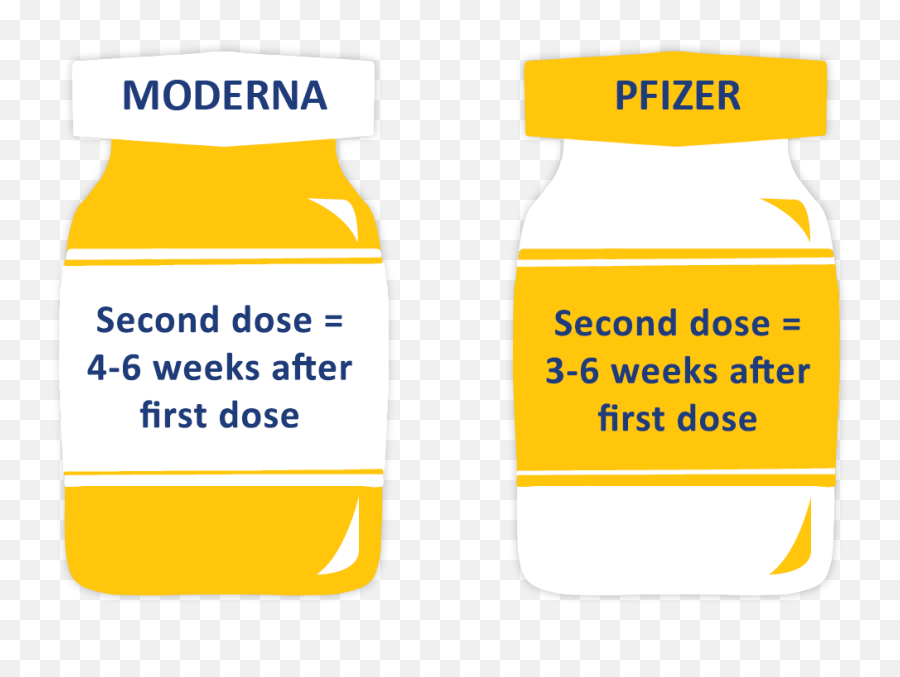
Those in remote communities, as part of broader community outreach vaccination programs.Aboriginal and Torres Strait Islander individuals.Individuals with specified medical conditions that increase their risk of severe.The Australian Technical Advisory Group on Immunisation (ATAGI) previously recommended vaccination using Comirnaty (Pfizer) for adolescents fromġ2 years of age that belong to the following groups 1: A two-dose schedule using Comirnaty (Pfizer) or Spikevax (Moderna) is recommended.Vaccination against COVID-19 is recommended for all individuals from 12 years of age, extending the current recommendation for those aged 16 years and older.This statement was originally published on 27 August 2021, and has now been updated. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
